 CRC Handbook of Chemistry and Physics (85th ed.). Half-life, spin, and isomer data selected from the following sources.International Union of Pure and Applied Chemistry. "News & Notices: Standard Atomic Weights Revised". 
"Atomic weights of the elements 2005 (IUPAC Technical Report)". Over a period of 2 years, 9 cross-check 226 were received from the Quality Assurance sigma control limits. de Laeter, John Robert Böhlke, John Karl De Bièvre, Paul Hidaka, Hiroshi Peiser, H. The expected precision for radium-226, based error, is 0.3 pCi/liter for samples up to 1.0 These are the values used in the Duplicate Quality Assurance Branch.Isotopic compositions and standard atomic masses from:.Finally, the exposure rate at one foot from one uCi of Ra-226 is approximately 10 uR/hr above background. Similarly, the exposure rate at one meter from one uCi of Ra-226 is approximately 1 uR/hr above background. Audi, Georges Bersillon, Olivier Blachot, Jean Wapstra, Aaldert Hendrik (2003), "The N UBASE evaluation of nuclear and decay properties", Nuclear Physics A, 729: 3–128, Bibcode: 2003NuPhA.729.3A, doi: 10.1016/j.nuclphysa.2003.11.001 Ra 226 is approximately 1 mR/hr Exposure Rate (X) 22-226 is approximately 1 mR/hr.^ Excluding those " classically stable" nuclides with half-lives significantly in excess of 232Th e.g., while 113mCd has a half-life of only fourteen years, that of 113Cd is nearly eight quadrillion years.^ This is the heaviest nuclide with a half-life of at least four years before the " sea of instability".No alpha activity attributable to the new isomer has been detected the alpha half-life is probably greater than 300. No growth of Cf 248 was detected, and a lower limit for the β − half-life can be set at about 10 4. This was ascribed to an isomer of Bk 248 with a half-life greater than 9. "The isotopic analyses disclosed a species of mass 248 in constant abundance in three samples analysed over a period of about 10 months. "The alpha half-life of berkelium-247 a new long-lived isomer of berkelium-248". ^ Specifically from thermal neutron fission of uranium-235, e.g.Radium's longest lived isotope, at 1,600 years, thus merits the element's inclusion here. While actually a sub-actinide, it immediately precedes actinium (89) and follows a three-element gap of instability after polonium (84) where no nuclides have half-lives of at least four years (the longest-lived nuclide in the gap is radon-222 with a half life of less than four days). 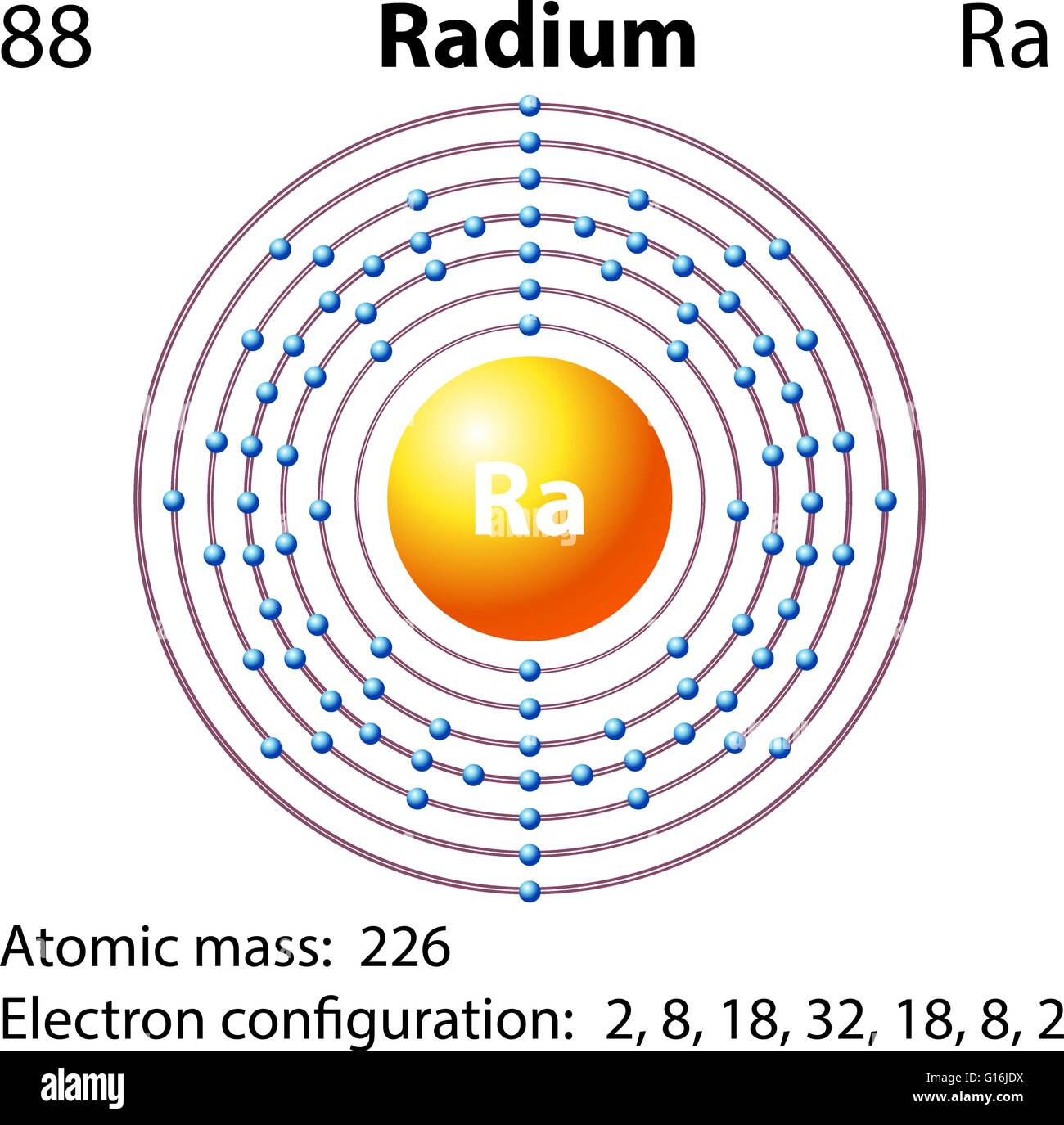
"α decay studies of very neutron-deficient francium and radium isotopes". ^ "First observations of short-lived pear-shaped atomic nuclei"."The NUBASE2020 evaluation of nuclear properties" (PDF). Radium forms when uranium and thorium break down in rocks and soil. This was the first discovery of an asymmetrical nucleus. What is RADIUM-226 &228 Radium is a radioactive metal found in nature. In 2013 it was discovered that the nucleus of radium-224 is pear-shaped. Radium has 34 known isotopes from 201Ra to 234Ra. 226Ra occurs in the decay chain of 238U (often referred to as the radium series). 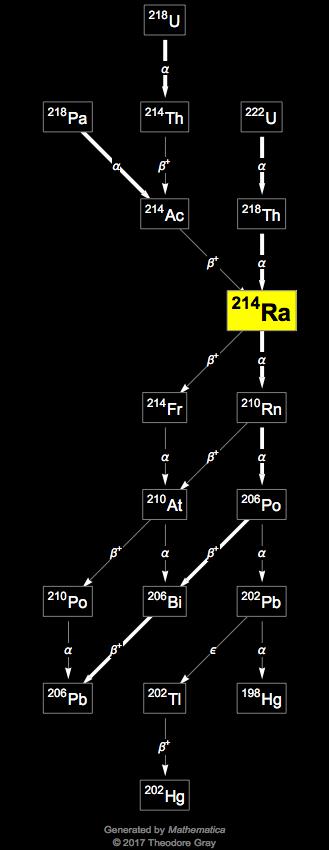
The longest lived, and most common, isotope of radium is 226Ra with a half-life of 1600 years. Radium ( 88Ra) has no stable or nearly stable isotopes, and thus a standard atomic weight cannot be given. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
